Polymers and Plastics
This page contains information on the following topics:
- Polymer Morphology
- Importance of morphology to Injection Molding
- Thermal Transitions Temperatures in Polymers
- Polymer Viscoelasticity
- Newtonian Fluid
- Non-Newtonian Fluid
- Plastics Density
- Plastics Shrinkage
- Melt Flow Rate or Melt Flow Index
Polymers
Polymers are large molecules whose molecular weight can range from the thousands to millions. Polymers are built up by the repetition of low molecular weight units. ‘poly’ means many and ‘mer’ = parts. For example: Polyethylene is made by ‘joining’ many ethylene molecules together. The process of ‘joining’ is called polymerization.

Polymer Morphology
Polymer morphology is the arrangement of the polymer molecules. There are two types of possible arrangements: Amorphous and Crystalline.

| Amorphous |
Crystalline |
| Random Structure | Presence of Ordered Structure – Crystallites |
| Broad Melt Processing Range | Narrow Melt Processing Range |
| Low Volumetric Shrinkage | High Volumetric Shrinkage |
| Lower Mechanical Properties | Higher Mechanical Properties |
| Transparent | Translucent or Opaque |
| Example – ABS, PS, ASA | Example – Polyesters, Nylons, Polyolefins |
Importance of morphology to Injection Molding
Depending on the morphology, the setting of the process parameters including the tolerances can both change. For example, right mold temperatures are extremely critical to help achieve the required properties in the molded part. However, in case of amorphous materials although important for properties, they play a bigger role for the cosmetics. Tests have shown the ABS parts molded at 65 deg F still retain the required strength for certain properties. In case of crystalline materials, higher screw speeds are required to melt the crystallites with the help of the shear energy. In case of amorphous materials high screw speeds are not a must. Melt homogeneity can be obtained at lower screw speeds.
Thermal Transitions Temperatures in Polymers
Thermal transitions temperatures are where the polymers change their state from one form to another just as H2O (not a polymer) is solid below 0 deg C, liquid between 0 and 100 deg C and gas above 100 deg C. In polymers there are three main types of such transitions temperatures.
The Glass Transition Temperatures – Tg
Below this temperature polymers are hard and brittle and above this the polymers are soft and flexible. In injection molded parts, post mold shrinkage will depend upon where the Tg is in relative to the room temperature and/or service temperature. If the Tg is above the room temperature and/or service temperature, post mold shrinkage is minimal. If the Tg is below room temperature and/or service temperature then post mold shrinkage will continue to take place till equilibrium is reached (think annealing).
During molding, if you set the mold tempertures too low – below the Tg, then you will not let the moecules reach equilibrium and the molded part will have a lot of in-built stress causing failures when a load is applied. The part can also change dimensions resulting in warpage and/or failures in a final assembly. In crstalline materials (see below) the Tc is always higher than the Tg and the mold temperatures should be set close to the TC.
The Melt Transition Temperature – Tm
This exists for crystalline materials only. Below this temperature the polymers are soft and flexible and above this the polymer is molten.
The Crystallization Temperature – Tc
This exists for crystalline materials only. When the melt is cooling down, the energy at this temperature is just sufficient to let the crystallites form. In molding this temperature dictates the required mold temperature range. If mold temperatures are set below these then the chances of the formation of crystallites is reduced thus affecting the properties.


Polymer Viscosity
The study of the viscosity and flow of polymers is called Rheology.
Viscoelasticity
When liquids are subjected to a force, the deformation is permanent. This property is called viscosity. Viscosity also is an indicator of the resistance to flow. Liquids are said to be viscous. When solids, example: metals, are subjected to a certain limited force the deformation is not permanent. When the force is released the solid recovers to its original shape. This property is called elasticity. The word ‘limited’ is used because excessive force causes permanent deformation, another topic of discussion. Plastics exhibit both a viscous component and an elastic component in their solid form and in their molten form. They are said to be viscoelastic and the behavior is called visoelasticity. This can be visualized with the spring and dash pot model.

Newtonian Fluid
A fluid whose viscosity is not influenced by the shear rate that is being applied to it. The viscosity remains constant as the shear changes. Shear rate in injection molding is synonymous to injection speed.
Non-Newtonian Fluid
A fluid whose viscosity changes with the change in the shear rate that is being applied to it. The viscosity does not remain constant as the shear changes.

All plastics are a combination of non – Newtonian and Newtonian behavior. At lower shear rates, the plastic is non-Newtonian but as the shear rate increases, the plastic tends to exhibit a Newtonian behavior. This happens because with increasing shear rate, the polymer molecules start to untangle from each other and start to align themselves in the direction of flow. After a certain shear rate most of the molecules are aligned in the direction of the flow and increasing the injection speed does not further affect the viscosity.

Important properties of Plastics
To understand Scientific Molding it is important to understand certain properties. They are as follows:
Density
Density of a material is the measure of the mass per unit volume and is generally expressed as grams per centimeter cube. Example: Density of General Purpose Polystyrene (GPPS) = 1.06 gms/cc. All molding barrels have their shot sizes expressed in terms of GPPS. For example, when a shot size is specified as 100 gms, it means that it can hold 100 grams of GPPS. If the molding material changes to 30% glass filled Nylon (density = 1.35 gms/cc) then the same barrel will now hold 125.5 gms of Nylon and so the shot size is now 125.5 gms.

Shrinkage
Shrinkage is the ratio of the volume of the plastic in the melt to the volume of the plastic in its final useable state. When applied to molding, it is expressed as the ratio of the dimension of the mold to the dimension of the part that is molded. Shrinkage is expressed in inches per inches or in percentage.
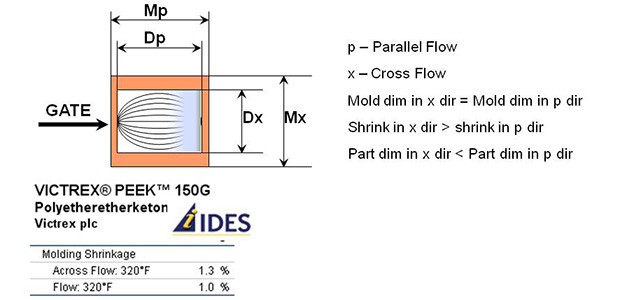
In case of crystalline materials, shrinkage is not the same in the cross flow and the parallel flow direction. Such materials are called anisotropic materials. In case of amorphous materials, shrinkage is the same in the cross flow and the parallel flow direction. Such materials are called isotropic materials.
Melt Flow Rate or Melt Flow Index
Melt Flow rate (MFR) or Melt Flow Index (MFI) is the amount of material extruded in 10 minutes under certain testing conditions. The plastic is fed into a heated vertical barrel and a known weight is kept on top of the plunger. The amount of plastic that is extruded in 10 minutes is called the MFR or MFI.

MFI and viscosity are inversely related:
- High Melt Viscosity = Low MFI
- Low Melt Viscosity = High MFI

